Class III Antiarrhythmics (Potassium Channel Blockers)
General Pharmacology
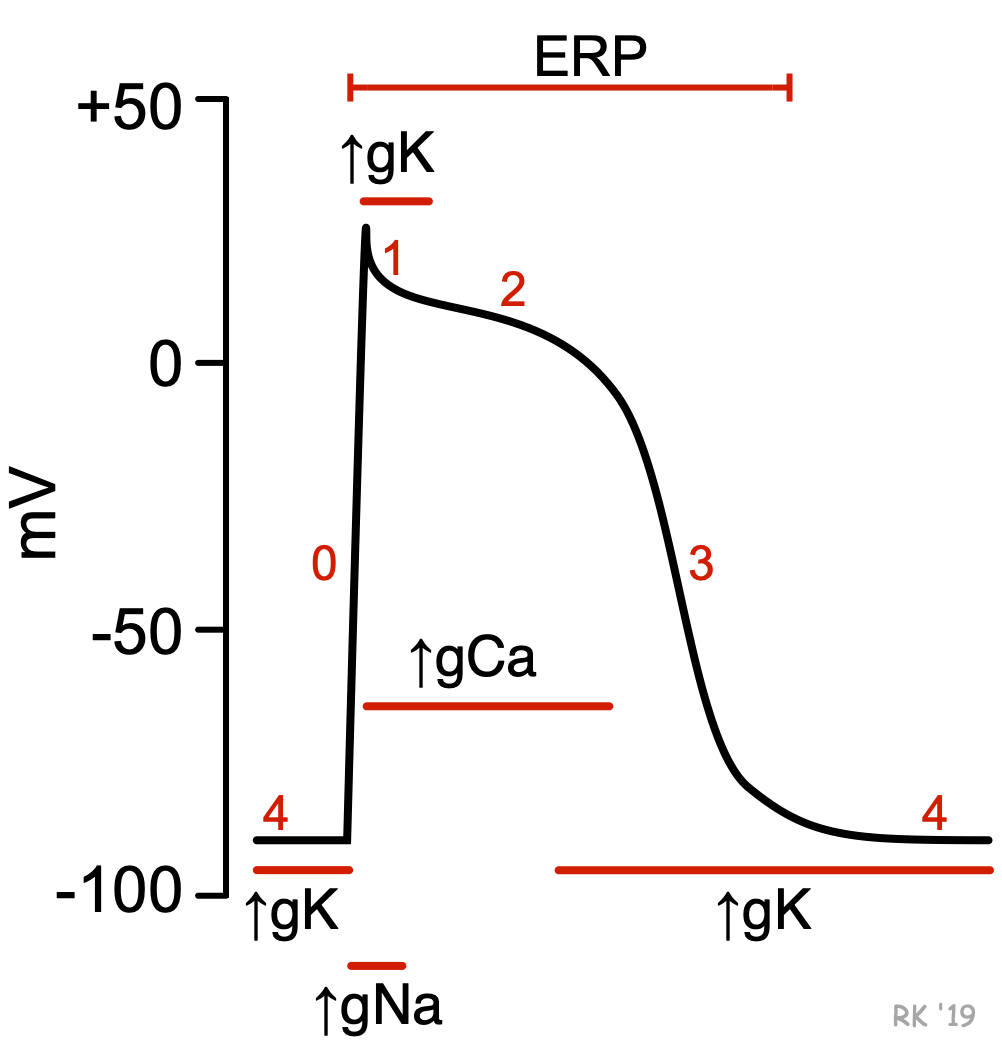 All class III antiarrhythmic drugs share a common electrophysiological mechanism in that they prolong the action potential duration, and most of the drugs in this class do so primarily by inhibiting repolarizing potassium currents. Therefore, these compounds are often referred to as “potassium channel blockers.” To understand this mechanism of these compounds, the following provides a brief review of cardiac action potential generation.
All class III antiarrhythmic drugs share a common electrophysiological mechanism in that they prolong the action potential duration, and most of the drugs in this class do so primarily by inhibiting repolarizing potassium currents. Therefore, these compounds are often referred to as “potassium channel blockers.” To understand this mechanism of these compounds, the following provides a brief review of cardiac action potential generation.
In non-nodal cardiac tissue, such as ventricular cardiomyocytes (see figure; click here for more details), action potentials are initiated when a cell is depolarized to a threshold potential by an adjacent cell. This leads to rapid opening of fast sodium channels and a slower opening of L-type calcium channels (increased sodium and calcium conductance; gNa and gCa, respectively) that permit sodium and calcium to enter the cell during phases 0–2. As these channels become inactivated, potassium channels open (increased gK) permitting potassium ions to leave the cell, which causes repolarization of the membrane potential (phases 3 and 4). Potassium channels remain open until the next action potential is triggered. A different type of potassium channel is responsible for the initial repolarization (phase 1). Potassium channels are also responsible for repolarizing slow-response action potentials in the sinoatrial and atrioventricular nodes.
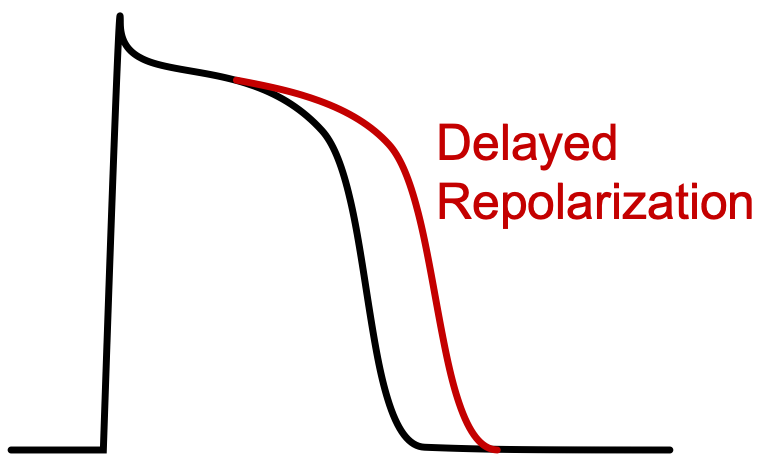 Class III antiarrhythmic compounds (Vaughan-Williams classification) bind to and block potassium channels that are responsible for phase 3 repolarization. By blocking these channels, action potential repolarization is delayed, which leads to an increase in action potential duration and an increase in the effective refractory period (ERP). Most of the class III drugs, however, have additional mechanisms of action that may contribute to their electrophysiological actions.
Class III antiarrhythmic compounds (Vaughan-Williams classification) bind to and block potassium channels that are responsible for phase 3 repolarization. By blocking these channels, action potential repolarization is delayed, which leads to an increase in action potential duration and an increase in the effective refractory period (ERP). Most of the class III drugs, however, have additional mechanisms of action that may contribute to their electrophysiological actions.
Prolongation of ventricular action potentials increases the QT interval in the electrocardiogram. This is a common effect of all Class III antiarrhythmic drugs. The electrophysiological changes prolong the length of time that the cell is unexcitable (refractory) and therefore make the cell less excitable.
By increasing the ERP, these drugs are very useful in suppressing tachyarrhythmias caused by reentry mechanisms. Reentry occurs when an action potential reemerges into normal tissue that is no longer refractory. When this happens, a new action potential is generated prematurely (before normal activation) and a circular, repeating pattern of early and rapid activation can develop, which leads to tachycardia. If the ERP of the normal tissue is lengthened, then the reemerging action potential may find the normal tissue refractory and premature activation will not occur.
Specific Drugs and Therapeutic Indications
The following table summarizes Class III compounds in terms of their therapeutic use and some special or distinguishing characteristics.
| Drug | Therapeutic Uses | Comments |
| amiodarone | ventricular tachycardia, including ventricular fibrillation; atrial fibrillation and flutter (off-label use) | very long half-life (25-60 days); Class I, II, III & IV actions and therefore decreases phase 4 slope and conduction velocity; side effects include (some serious): pulmonary fibrosis; hypothyroidism; hepatotoxicity; corneal micro-deposits, skin discoloration |
| dronedarone | atrial fibrillation (non-permanent) and flutter | structurally related (but non-iodinated) to amiodarone, but has a much smaller volume of distribution and shorter elimination half-life (~24 hr); Class I, II, III & IV actions; contraindicated in severe or recently decompensated, symptomatic heart failure; although less toxic than amiodarone, it is less efficacious in atrial fibrillation |
| bretylium | life-threatening ventricular tachycardia and fibrillation | IV only; initial sympathomimetic effect (norepinephrine release) followed by inhibition, which can lead to hypotension |
| sotalol | ventricular tachycardia; atrial flutter and fibrillation | inhibits opening of repolarizing K+ channels and increases ERP; also has Class II (beta-blocker) activity and therefore slows sinus rate |
| ibutilide | atrial flutter and fibrillation (acute termination) | activates slow inward Na+ currents during early phase 3 and inhibits opening of repolarizing K+ channels, which delays repolarization and increases ERP; IV only; can cause life-threatening ventricular arrhythmias; infrequent non-cardiac side effects |
| dofetilide | atrial flutter and fibrillation; paroxysmal supraventricular tachycardia (off-label) | approved for acute atrial flutter and fibrillation; very selective K+-channel blocker; can cause life-threatening ventricular arrhythmias |
Abbreviations: IV, intravenous.
Cardiac Side Effects and Contraindications
These drugs, like Class I drugs, are proarrhythmic as well as being antiarrhythmic. For example, the increase in action potential duration can produce torsades de pointes (a type of ventricular tachycardia), especially in patients with long-QT syndrome. Amiodarone, because of its Class IV effects, can cause bradycardia and atrioventricular block, and therefore is contraindicated in patients with heart block or sinoatrial node dysfunction.
Revised 11/30/2023
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)