The Pharmacologic Treatment of Heart Failure cont.
- Page 1: Causes of Heart Failure
- Page 2: Pathophysiology of Heart Failure
- THIS PAGE: Rationale for Drug Therapy
- Page 4: Classes of Drugs Used to Treat Heart Failure
Rationale for Drug Therapy
The goal of drug therapy in heart failure is to improve cardiac function and reduce the clinical symptoms associated with heart failure (e.g., edema, shortness of breath, exercise intolerance). Improving cardiac function, along with reducing blood volume, can dramatically improve the clinical symptoms. The treatment of heart failure caused by systolic dysfunction follows clear clinical guidelines based upon many clinical trials. Diastolic dysfunction, however, is more difficult to treat and there is no clear consensus regarding the best therapeutic options apart from targeting clinical symptoms related to fluid retention.
Systolic Dysfunction
Systolic dysfunction results from a loss of intrinsic contractility and is usually associated with a dilated ventricle. A decrease in stroke volume coupled to an increase in ventricular end-diastolic volume leads to a significant reduction in ejection fraction (EF). Normally, EF is greater than 55%. In severe systolic dysfunction, the EF may be less than 20%. This condition is commonly referred to as heart failure with reduced ejection fraction (HFrEF). An example of systolic dysfunction is dilated cardiomyopathy (DCM), which can result from known or unknown diseases that impair ventricular function. With systolic dysfunction, the Frank-Starling curves shifts down and to the right because of the loss of contractility (see figure: shift from point A to B). When this occurs, stroke volume is reduced and preload (LVEDP in figure) is increased secondarily. Compensatory increases in blood volume further increase preload and dilate the ventricle.
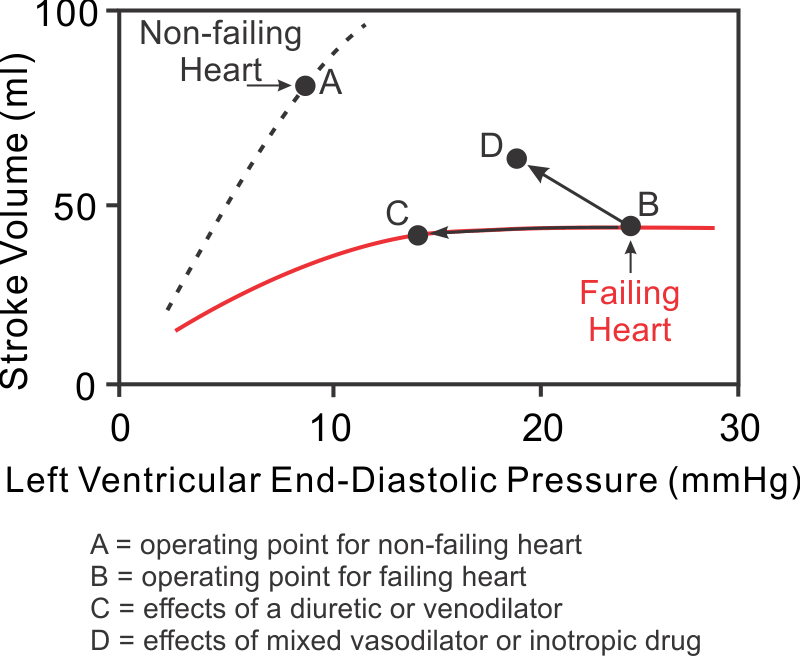 Ventricular stroke volume can be improved by increasing preload, decreasing afterload, and increasing inotropy. In HFrEF, preload is already elevated because of ventricular dilation resulting from ventricular remodeling and increased blood volume. Increasing the preload further in the presence of HFrEF does not necessarily increase stroke volume because the ventricle is usually functioning on the flat, depressed region of the Frank-Starling curve (see region between points C and B in figure). Furthermore, increasing preload will exacerbate pulmonary or systemic congestion and edema, which occurs when end-diastolic pressure exceeds 20 mmHg. Therefore, increasing preload is not a viable option for increasing cardiac output in heart failure patients.
Ventricular stroke volume can be improved by increasing preload, decreasing afterload, and increasing inotropy. In HFrEF, preload is already elevated because of ventricular dilation resulting from ventricular remodeling and increased blood volume. Increasing the preload further in the presence of HFrEF does not necessarily increase stroke volume because the ventricle is usually functioning on the flat, depressed region of the Frank-Starling curve (see region between points C and B in figure). Furthermore, increasing preload will exacerbate pulmonary or systemic congestion and edema, which occurs when end-diastolic pressure exceeds 20 mmHg. Therefore, increasing preload is not a viable option for increasing cardiac output in heart failure patients.
Decreasing afterload with vasodilator drugs significantly enhances ventricular stroke volume (figure: B→D) and ejection fraction in failing hearts. Reducing afterload increases ejection velocity (see force-velocity relationship), which augments the stroke volume. Therefore, reducing afterload is very effective in treating HFrEF because it increases stroke volume and decreases preload, which improves EF.
Increasing inotropy also increases stroke volume and ejection fraction, and reduces preload (figure: B→D); however, most positive inotropic drugs (e.g., sympathomimetics and phosphodiesterase inhibitors) should only be used for acute systolic failure or end stage failure because prolonged use of these drugs has been shown to worsen the outcome and increase mortality. The short-term benefit of such drugs is that they increase stroke volume with a modest reduction in preload, all of which are beneficial. However, inotropic drugs increase oxygen demand, which is deleterious with long-term use.
Decreasing afterload and increasing inotropy both reduce ventricular end-systolic volume, which causes the end-diastolic volume to decrease secondarily. Reduced venous pressure decreases capillary pressure and fluid filtration in the lungs (left-sided failure) and systemic tissues (right-sided failure), diminishing edema.
Diuretic drugs are used in most heart failure patients because heart failure leads to renal retention of sodium and water, which increases blood volume and venous pressures. These changes promote vascular congestion and edema formation. Pulmonary edema, which occurs with left ventricular failure, can be life-threatening because pulmonary oxygen exchange is compromised. Diuretics promote renal loss of sodium and water and, therefore, are very effective in reducing vascular congestion and edema. In fact, most HFrEF patients are treated with a diuretic besides other drugs, such as vasodilators or cardiostimulatory drugs. Although diuretics reduce ventricular preload (figure: B→C), this usually does not significantly reduce stroke volume because the depressed Frank-Starling curve in systolic dysfunction is relatively flat at high preload volumes and pressures.
Another drug used in some patients with systolic heart failure is ivabradine. This relatively new drug blocks sinoatrial "funny" currents that generate pacemaker currents controlling heart rate. By blocking these currents, ivabradine reduces heart rate and myocardial oxygen demand, which clinical trials have shown to be beneficial in heart failure patients. Although beta-blockers also reduce heart rate, their actions on beta-adrenoceptors can also depress inotropy. Therefore, ivabradine acts as a "pure" heart rate reducing drug.
Diastolic Dysfunction
This type of ventricular failure is related to impaired ventricular filling caused by hypertrophied (less compliant) ventricles or by impaired ventricular relaxation. Hypertrophy can result from chronic hypertension or aortic valve stenosis. Some patients may have a genetic defect that causes hypertrophic cardiomyopathy (HCM). Diastolic dysfunction can also occur due to the stiffening of the ventricular wall (restrictive cardiomyopathy) caused by fibrosis. These patients will often have normal or near normal ejection fractions, and therefore this type of failure is referred to as heart failure with preserved ejection fraction (HFpEF). Diastolic dysfunction results in large increases in ventricular end-diastolic pressure, which can lead to pulmonary edema. Despite a large end-diastolic pressure, the end-diastolic volume may actually be reduced because of the decreased ventricular compliance.
HFpEF is more difficult to treat than systolic dysfunction. If there is pulmonary edema, diuretics are given; however, they are given cautiously because removing too much volume can significantly reduce end-diastolic volume and therefore stroke volume in a stiff ventricle. A patient may be given calcium-channel blockers such as verapamil and diltiazem. These drugs are contraindicated in HFrEF because they reduce inotropy and stroke volume, but inotropy may be normal in diastolic dysfunction, so these drugs do not seriously impair stroke volume in these patients. Calcium-channel blockers seem to have their benefit by improving ventricular relaxation and reducing heart rate (which permits more time for filling). They also promote regression of cardiac hypertrophy, reduce arterial pressure and improve coronary blood flow. Beta-blockers help these patients by promoting regression of hypertrophy, reducing arterial pressure, slowing heart rate, and reducing inotropy. The negative inotropic effects of both calcium-channel blockers and beta-blockers are useful in patients with HCM that also have outflow obstruction, which can be caused by thickening of the interventricular septum and by an abnormal motion of the anterior leaflet of the mitral valve. ACE inhibitors may also be used because of their beneficial effect on ventricular remodeling and arterial pressure. Cardiostimulatory drugs (sympathomimetics and digitalis compounds) are not recommended for treating diastolic dysfunction, particularly in patients with obstructive HCM because increasing inotropy can cause increased outflow obstruction and higher ventricular systolic pressures.
It is important to note that while pharmacologic intervention can improve the clinical status of heart failure patients, cardiac function and organ perfusion are not restored to normal values. In the late stages of chronic failure or in severe acute failure, a patient may become refractory to drug therapy. When this occurs, the only option is surgical correction of the underlying problem (if identified), mechanical cardiac assist devices, or heart transplant.
Go to Next Page
Classes of Drugs Used to Treat Heart Failure
Revised 11/30/2023
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)