Adenosine
General Pharmacology
Adenosine is a naturally occurring purine nucleoside that forms from the breakdown of adenosine triphosphate (ATP). ATP is the primary energy source in cells for transport systems and many enzymes. Most ATP is hydrolyzed to ADP, which can be further dephosphorylated to AMP. Most ADP and AMP that form in the cell are rephosphorylated by the mitochondria through enzymatic reactions requiring oxygen. If large amounts of ATP are hydrolyzed, and especially if there is insufficient oxygen available (i.e., hypoxia), then some of the AMP can be dephosphorylated to adenosine by the cell membrane associated enzyme, 5'-nucleotidase.
Adenosine has a short half-life. In human blood, its half-life is less than 10 seconds. There are two important metabolic fates for adenosine.
- Adenosine is rapidly transported into red blood cells (and other cell types) where it is rapidly deaminated by adenosine deaminase to inosine, which is further broken down to hypoxanthine, xanthine, and uric acid which is excreted by the kidneys. Adenosine deamination also occurs in plasma, but at a lower rate than that which occurs within cells. Dipyridamole is a vasodilator drug that blocks adenosine uptake by cells, reducing the metabolism of adenosine. Therefore, one significant mechanism for dipyridamole-induced vasodilation is its potentiation of extracellular adenosine.
- Adenosine can be acted on by adenosine kinase and rephosphorylated to AMP. This salvage pathway helps maintain the adenine nucleotide pool in cells.
Cardiac electrical effects
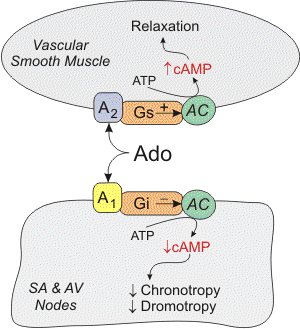 In cardiac nodal tissue, adenosine binds to type 1 (A1) receptors, which are coupled to Gi-proteins. Activation of this pathway opens potassium channels, which hyperpolarizes the cell. Activation of the Gi-protein also decreases cAMP, which inhibits L-type calcium channels and therefore calcium entry into the cell. In cardiac pacemaker cells in the sinoatrial node, adenosine acting through A1 receptors inhibits the pacemaker current (If), which decreases the slope of phase 4 of the pacemaker action potential and decreases its spontaneous firing rate (negative chronotropy). Inhibition of L-type calcium channels by adenosine also decreases conduction velocity (negative dromotropic effect) at the atrioventricular (AV) node. Finally, adenosine, by acting on presynaptic purinergic receptors on sympathetic nerve terminals, inhibits the release of norepinephrine. In terms of its electrical effects in the heart, adenosine decreases the heart rate at the SA node and reduces conduction velocity at the AV node. The latter effect can produce atrioventricular block. Note, however, that when adenosine is infused systemically into humans, the heart rate can increase because of baroreceptor reflexes activated by systemic vasodilation and hypotension.
In cardiac nodal tissue, adenosine binds to type 1 (A1) receptors, which are coupled to Gi-proteins. Activation of this pathway opens potassium channels, which hyperpolarizes the cell. Activation of the Gi-protein also decreases cAMP, which inhibits L-type calcium channels and therefore calcium entry into the cell. In cardiac pacemaker cells in the sinoatrial node, adenosine acting through A1 receptors inhibits the pacemaker current (If), which decreases the slope of phase 4 of the pacemaker action potential and decreases its spontaneous firing rate (negative chronotropy). Inhibition of L-type calcium channels by adenosine also decreases conduction velocity (negative dromotropic effect) at the atrioventricular (AV) node. Finally, adenosine, by acting on presynaptic purinergic receptors on sympathetic nerve terminals, inhibits the release of norepinephrine. In terms of its electrical effects in the heart, adenosine decreases the heart rate at the SA node and reduces conduction velocity at the AV node. The latter effect can produce atrioventricular block. Note, however, that when adenosine is infused systemically into humans, the heart rate can increase because of baroreceptor reflexes activated by systemic vasodilation and hypotension.
Vasodilator actions
Adenosine binds to purinergic receptors in different cell types, where it has several actions. One important action is vascular smooth muscle relaxation, which leads to vasodilation. This is an important mechanism for matching coronary blood flow to the metabolic needs of the heart. In coronary vascular smooth muscle, adenosine binds to adenosine type 2A (A2A) receptors, which are coupled to the Gs-protein. Activation of this G-protein stimulates adenylyl cyclase (AC in figure), increases cAMP, and causes protein kinase activation. This activates KATP channels, which hyperpolarizes the smooth muscle, causing relaxation. Increased cAMP also causes smooth muscle relaxation by inhibiting myosin light chain kinase, which leads to decreased myosin phosphorylation and a decrease in contractile force. There is also evidence that adenosine inhibits calcium entry into the cell through L-type calcium channels. Since calcium regulates smooth muscle contraction, reduced intracellular calcium causes relaxation. In some types of blood vessels, there is evidence that adenosine produces vasodilation through increases in cGMP, which leads to inhibition of calcium entry into the cells, opening of potassium channels, and activation of myosin light chain phosphatase.
Therapeutic and Diagnostic Use
The major therapeutic use of adenosine is as an antiarrhythmic drug for the rapid treatment of supraventricular tachycardia caused by AV nodal reentry by suppressing AV nodal conduction. For these indications, adenosine is administered either as a bolus intravenous injection or as an intravenous infusion. Adenosine is not effective for atrial flutter or fibrillation.
Although adenosine is a powerful vasodilator, especially in coronary circulation, it is not used as a vasodilator to treat coronary artery disease. The reason is that it is short-acting, limited to intravascular administration, and can produce coronary vascular steal. When administered by intravenous infusion, it can produce hypotension and atrioventricular block.
Adenosine's unique vasodilator properties, however, are utilized in cardiac imaging during stress tests to determine coronary fractional flow reserve (a measure of severity of coronary stenosis), and to assess pulmonary vasodilator responses in patients with pulmonary hypertension.
Side effects
Most of adenosine's side effects are related to its vasodilator properties. Patients can experience flushing and headaches, both of which are related to vasodilation. Adenosine can produce rapid arterial hypotension; however, this is reversed shortly after stopping the infusion of adenosine because of its short half-life. Coronary vascular steal is of theoretical concern in some patients with coronary artery disease, although there is no clinical evidence supporting this adverse effect. Methylxanthines such as caffeine and theophylline competitively antagonize the binding of adenosine at its purinergic receptor. Finally, adenosine may produce undesirable AV block; however, this is usually rapidly corrected by stopping adenosine administration. Therefore, adenosine is contraindicated in patients with preexisting second or third degree AV block.
Revised 11/29/23
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)